Complete your application#
Last modified: 02 Dec 2025
Your full application comprises up to three sections:
application form
data request
codelist (if you are applying to access certain NHS England datasets - see below)
1. Complete your application form#
Log into UK LLC Apply and complete the following sections (you can save your application and come back to it later):
Project overview
Methodology
Benefits and public involvement
Funding
Lawful basis
Ethics
Data
Setting
Outputs.
A pdf of the application form is available here.
In particular, please take into consideration these following key points:
a) Co-applicants#
You can invite co-applicants to work with you on your application, but these should only be researchers who will be accessing data in the UK LLC TRE. If you deem it absolutely necessary to invite a co-applicant to help you develop your application, but this person won’t be accessing the TRE, please restrict these co-applicants to one or two at the most.
There can be only one main applicant per application
Only the main applicant can submit the application
A PhD student can not be the main applicant.
b) Ethics#
You must have obtained a favourable opinion from an independent Research Ethics Committee (REC) if you are applying to access data of elevated sensitivity and/or if you are using novel or contentious research methods (see definitions below). We accept faculty ethics committee or Health Research Authority REC approval. If you do not have a favourable opinion from an independent REC, you will be asked to fill in the Ethics Self-assesment Tool developed by the UK Statistics Authority. The form can be downloaded from within UK LLC Apply. Once completed, please upload your form to the Ethics section of your application. Your ethical assessment will be reviewed and, depending on the outcome, you may need to contact your institution’s REC for further advice - see the UK LLC ethics decision-making tree below.
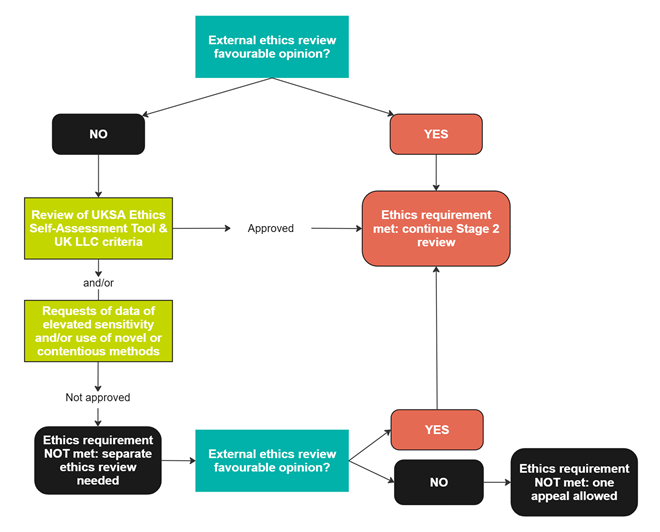
Figure 1 An overview of the UK LLC ethics decision-making process
Data of elevated sensitivity:
mental health
sexual health
drug and alcohol misuse
assisted pregnancy, termination of pregnancy, pregnancy in age <16 years
abuse
self-harm
suicide.
Novel or contentious research methods:
entirely novel or not commonly used methods
any artificial intelligence (AI) methods. (See UK LLC’s AI policy here.)
c) Public and Participant Involvement and Engagement (PPIE)#
All applications approved by UK LLC must be for the public good. You can find the definition for public good here. We strongly encourage you to think about and develop PPIE plans that cover the duration of your project (from developing research ideas to discussing results and dissemination). This is obligatory for projects involving data of elevated sensitivity and novel/contentious research methods. To understand how UK LLC assesses public good, please see UK LLC Data Access Public Review Panel Public Good Considerations.
2. Complete your data request#
Go to UK LLC Explore to build your data request. Select all the datasets you require by ticking a box next to the dataset name. All datasets are clustered according to the schema to which they belong, e.g. PLACE refers to place-based datasets, MCS refers to Millennium Cohort Study datasets. Then go to Selection and press Save - this will download a data_selection.csv file which you then upload in the Data section of your application.
3. Complete your codelist#
If you have selected any of the following six NHS England datasets you must complete a codelist:
CANCER (Cancer Registrations)
GDPPR (General Practice Extraction Service (GPES) Data for Pandemic Planning and Research)
HESAE (Accident & Emergency)
HESAPC (Admitted Patient Care)
HESOP (Outpatients)
PCM (Primary Care Medicines).
(HES: Hospital Episode Statistics)
To download the UK LLC codelist template and for guidance on completing your codelist, please see the Codelists and NHS England data guide. When you’ve completed your codelist, upload it in the Data section of your application.
Application review#
Your application will undergo internal and external review. You will be notified of the progress of your application - please see the How your application is reviewed guide for further information.
